Most of us in the radiation oncology world are familiar with treating a patient with a pacemaker or defibrillator, knowing that we need to minimize dose to the device and keep it out of the primary beam if possible. But the uncertainty in the risk to these implanted cardiac devices mean that the guidelines and procedures to care for these patients are not entirely straight forward. An old AAPM task group report (TG-34) from 1994 did a good job of explaining potential pacemaker malfunctions and recommending keeping pacemaker devices to no more than 2 Gy during radiation therapy. Alas, a lot has changed since 1994, including in the pacemaker world. Now it is increasingly common to encounter both implantable cardiac pacemakers (ICPs) and implantable cardioverter-defibrillators (ICDs) in the clinic.
These modern devices also have newer technology than those the old TG-34 recommendations were based from. Each manufacturer had their own recommendations and safety guidance, which varies widely. This all added up to a distinct void in up-to-date recommendations on how to best deal with these cardiac implantable electronic devices (CIEDs). That is where the AAPM Task Group report 203 (TG-203; 2019) comes in.
TG-203 does an excellent job of reviewing published literature evaluating effects of radiation therapy on CIEDs, explaining their function, design, and sensitivities, and defining how different factors in a radiation oncology clinic can affect the CIEDs. All of this thorough explanation sets up the key component of the report: recommendations on how to assess dose levels, estimate risks, and manage patients with CIEDs. For detailed reading on all things CIEDs and radiation therapy, I refer you to TG-203. Here, we will take a quick look at recommendations from the report and how to implement them in your clinic.
There are three main processes associated with care of patients with CIEDs:
These modern devices also have newer technology than those the old TG-34 recommendations were based from. Each manufacturer had their own recommendations and safety guidance, which varies widely. This all added up to a distinct void in up-to-date recommendations on how to best deal with these cardiac implantable electronic devices (CIEDs). That is where the AAPM Task Group report 203 (TG-203; 2019) comes in.
TG-203 does an excellent job of reviewing published literature evaluating effects of radiation therapy on CIEDs, explaining their function, design, and sensitivities, and defining how different factors in a radiation oncology clinic can affect the CIEDs. All of this thorough explanation sets up the key component of the report: recommendations on how to assess dose levels, estimate risks, and manage patients with CIEDs. For detailed reading on all things CIEDs and radiation therapy, I refer you to TG-203. Here, we will take a quick look at recommendations from the report and how to implement them in your clinic.
There are three main processes associated with care of patients with CIEDs:
- Staff responsibilities for management of these patients
- Determining (and minimizing) the patient risk category
- Required action based on patient risk category
1. Staff responsibilities for management of patients with CIEDS
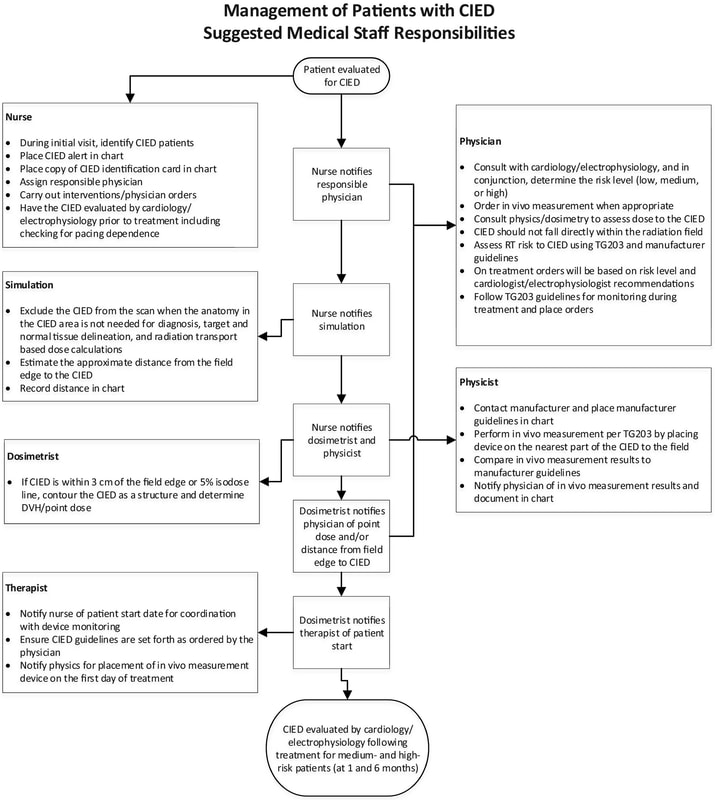
TG-203 Figure 6 shows a suggested workflow for the management of patients with CIEDs. Management involves coordination between nursing, physician, physics, dosimetry, and therapists. At a high level, nursing identifies CIED patients and initiates the process to have the physician consult with cardiology and place relevant orders (device interrogation, in vivo measurement, etc.), inform simulation to exclude the CIED if possible, notify dosimetry and physics to determine optimal planning for patient case and make proper measurements/estimations to determine dose level, and dosimetrist notifies therapists of patient start and to follow CIED guidelines for imaging/treatment.
TG-203 Figure 6 shows a suggested workflow for the management of patients with CIEDs. Management involves coordination between nursing, physician, physics, dosimetry, and therapists. At a high level, nursing identifies CIED patients and initiates the process to have the physician consult with cardiology and place relevant orders (device interrogation, in vivo measurement, etc.), inform simulation to exclude the CIED if possible, notify dosimetry and physics to determine optimal planning for patient case and make proper measurements/estimations to determine dose level, and dosimetrist notifies therapists of patient start and to follow CIED guidelines for imaging/treatment.
2. Determining (and minimizing) patient risk category TG-203
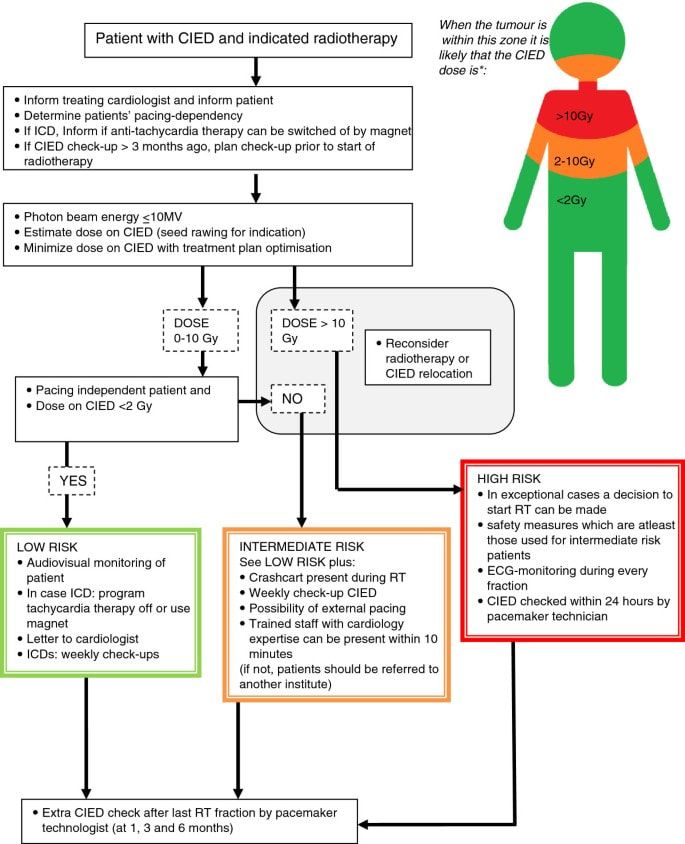
Figure 5 is a straightforward flowchart detailing how to determine risk level. Depending on treatment parameters (energy, field edge proximity, estimated dose to any part of the device, etc.), the patient will fall into one of three risk categories:
i. Low-Risk
ii. Medium-Risk
iii. High-Risk
I have found this flowchart to be incredibly useful as we implement updated policies and procedures in our clinic. It provides clear parameters and decision points for determining risk to patients with CIEDs.
Figure 5 is a straightforward flowchart detailing how to determine risk level. Depending on treatment parameters (energy, field edge proximity, estimated dose to any part of the device, etc.), the patient will fall into one of three risk categories:
i. Low-Risk
ii. Medium-Risk
iii. High-Risk
I have found this flowchart to be incredibly useful as we implement updated policies and procedures in our clinic. It provides clear parameters and decision points for determining risk to patients with CIEDs.
3. Required action based on patient risk category
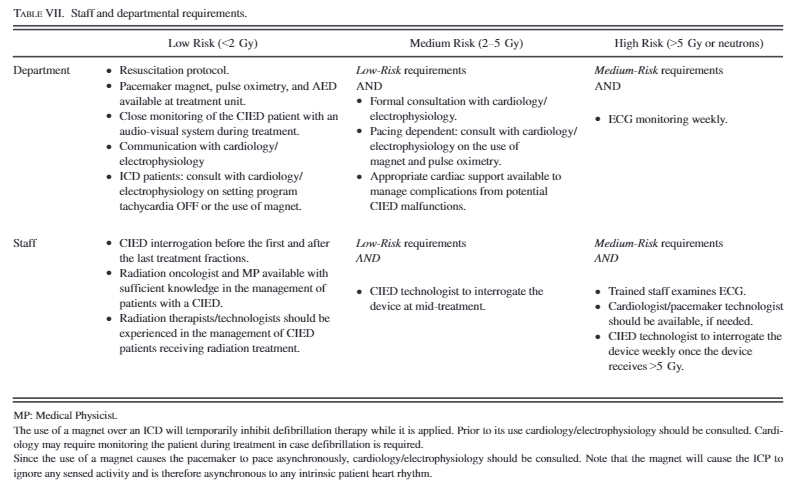
TG-203 Table VII outlines the recommended actions the department and staff take for CIED patients in each risk category. Departmental requirements include having proper protocols and equipment to both manage patients with CIED and care for a patient in case of device failure. Staff requirements include proper training and action taken to minimize risk of device failure and the potential harm a device failure could cause.
TG-203 Table VII outlines the recommended actions the department and staff take for CIED patients in each risk category. Departmental requirements include having proper protocols and equipment to both manage patients with CIED and care for a patient in case of device failure. Staff requirements include proper training and action taken to minimize risk of device failure and the potential harm a device failure could cause.
TG-203 is a refreshing update to TG-34 in caring for patients with CIEDs. With its inclusion and direction on a wide spectrum of possible clinical encounters of patients with CIEDs, following TG-203 guidelines in your clinic will contribute to improved quality and safety for your patients.
Austin Hadley, DMP